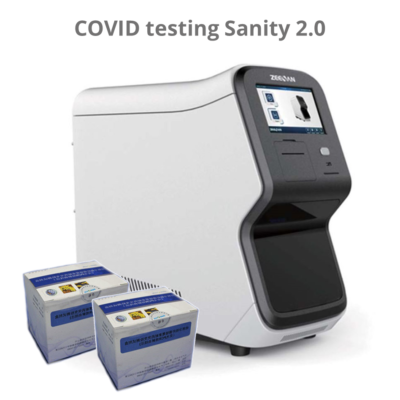
Consumables Sanity 2.0 – SARS-CoV-2 test kit (24 tests)
€ 223,85 € 185,00 ex. VAT
This kit is used for in vitro qualitative detection of novel coronavirus (SARS-CoV-2) ORF1ab and N gene in specimens, including nasopharyngeal swab, oropharyngeal swab, saliva, sputum, bronchoalveolar lavage fluid, for diagnosis of the COVID-19 infection, the suspected cluster cases and of the patients who need differential diagnosis of COVID-19 infection.
Available on backorder
This kit is used for in vitro qualitative detection of novel coronavirus (SARS-CoV-2) ORF1ab and N gene in specimens, including nasopharyngeal swab, oropharyngeal swab, saliva, sputum, bronchoalveolar lavage fluid, for diagnosis of the COVID-19 infection, the suspected cluster cases and of the patients who need differential diagnosis of COVID-19 infection.
The kit should be operated on Automatic Medical PCR Analysis System Sanity 2.0 System; Xiamen Zeesan Biotech Co.,
Ltd.) which has FAM, HEX, ROX and CY5 detection channels, in combination with the pathogen nucleid acid extraction kit.
Technical details:
- Repeatability & precision: the coefficient of variation (CV) of the Ct values is no higher than 5%.
- Specificity: no cross reactions were found
- Limit of detection (LOD): 200 copies/mL
About the Sanity 2.0:
The Automatic Medical PCR Analysis System Sanity 2.0 is an automated Point-of-Care (POC) system that integrates specimen extraction and purification, PCR amplification, signal generation and optical detection. Once the user loads the specimen into the extraction loading well and starts the run, all other operations are automated on the instrument. The software preloaded in the instrument will collect and analyze data, and automatically generate interpretation for test reports at the end of the run. The system utilizes magnetic particle technology for automated isolation and purification of nucleic acid from saliva, bronchoalveolar lavage fluid (BALF), sputum, swabs, etc.
The product is intended to be used by professional users only.
The Automatic Medical POC PCR Analysis System Sanity 2.0 performs all steps of the sample preparation procedure for 4 samples in a single run.
SARS-CoV-2 mutation panel kits:
Please note that we also have the following SARS-CoV-2 mutation panel kits available for usage on the Sanity 2.0:
- Sanity SARS-CoV-2 Mutation Panel kit (SKU: 331107)
- Sanity SARS-CoV-2 Variant P.1 Identification kit (SKU: 331103)
- Sanity SARS-CoV-2 Variant B.1.617 Identification kit (SKU: 331104)
- Sanity SARS-CoV-2 Variant B.1.351 Identification kit (SKU: 331105)
- Sanity SARS-CoV-2 Variant B.1.1.7. Identification kit (SKU: 331106)
More information about these kits is available on request, please send us an e-mail (info@prodiag.nl).
Storage conditions:
The kit should be stored at 2-8℃ in the dark. Production date and expiration date are shown on the package label.